Thesis: A continuous microfluidic dehydration process generates syringeable, semi-solid antibody formulations at ~360 mg/mL without centrifugation, addressing a major manufacturing barrier for high-dose subcutaneous delivery, while in vivo performance, regulatory pathways, and large-scale implementation remain open questions.
Executive summary – what changed and why it matters
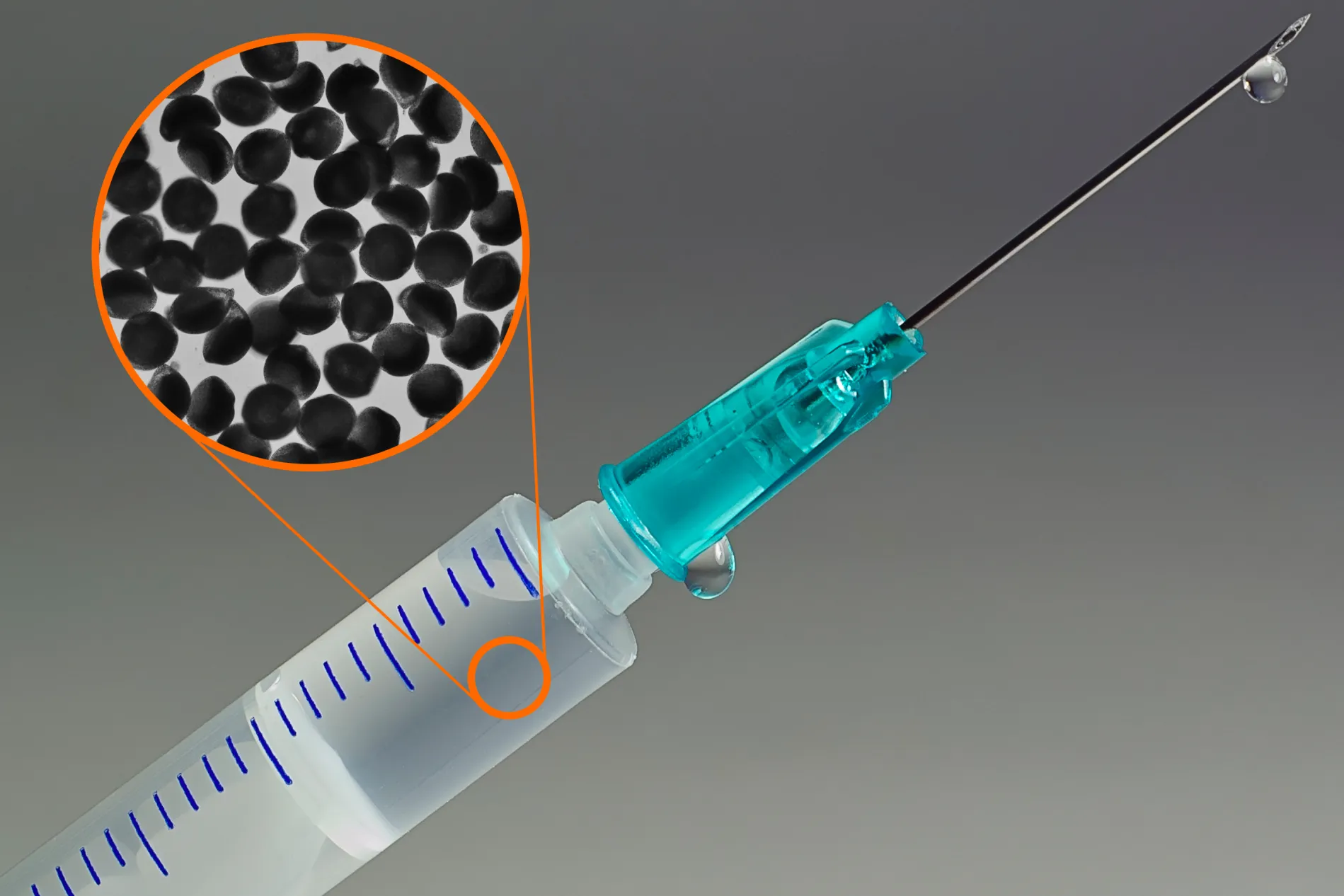
A team at MIT has converted monoclonal antibodies into ~100 µm alginate hydrogel particles using a continuous microfluidic solvent-dehydration step in pentanol. This approach concentrates antibodies to ~360 mg/mL, enabling more than 700 mg to fit in a 2 mL syringe with plunger forces below 20 N, and achieves stable formulations under refrigeration for at least four months. Crucially, it omits the centrifugation step central to a 2023 method, removing a key obstacle to continuous, GMP-compatible scale-up.
Technical overview – microfluidic dehydration and particle formation
The process begins with a water-in-oil emulsion formed at a microfluidic cross-junction, encapsulating an aqueous phase of antibody dissolved in alginate prepolymer. As droplets traverse a pentanol bath, water is extracted, driving antibodies into an amorphous solid dispersion (ASD) within the polymer matrix. Subsequent calcium cross-linking stabilizes the alginate shell. The resulting particles, mostly ~100 µm in diameter (range 60–200 µm), are collected and resuspended in a polyethylene glycol (PEG)–stabilized buffer suitable for injection.
Key performance metrics reported include:
- Final antibody concentration of ~360 mg/mL (6× increase over typical liquid formulations).
- Encapsulation efficiency exceeding 98% from a 60 mg/mL feed solution.
- Syringeability assessed by peak plunger force <20 N for a 2 mL load—well below conventional 40–50 N thresholds.
- Refrigerated stability for at least four months without loss of syringeability or observable aggregation.
By removing centrifugation—a batch-oriented step that concentrates and recovers particles—this route configures antibody encapsulation as a continuous, inline process, pointing toward more scalable manufacturing.
Contextual backdrop – breaking free from the 2023 bottleneck
In 2023, the same research lab demonstrated a bead/crystallization strategy to reach high antibody concentrations in alginate, but that workflow relied on centrifugation to recover crystalline beads—an operation challenging to parallelize and validate under GMP. The new solvent-dehydration route replaces both crystallization and centrifugation with a single continuous operation, tightening the link between droplet formation and final syringe-ready suspension.

This timing aligns with payer and provider demands to shift monoclonal antibody administration from infusion centers to subcutaneous or at-home injections, aiming to reduce healthcare costs, expand patient autonomy, and improve access. Removing a core manufacturing impediment could accelerate that transition—provided remaining unknowns are addressed.
Positioning among subcutaneous delivery approaches
Current high-dose subcutaneous strategies include viscosity-reducing excipients, hyaluronidase co-administration to enlarge injection volumes, and prefilled syringes loaded with concentrated liquid formulations (generally limited to ~150–200 mg/mL). Device-based innovations—solid implants or on-body infusers—offer alternative paths but often involve complex assemblies or compromise patient comfort.
The microfluidic hydrogel particle approach diverges by packaging antibodies in a semi-solid, ASD-laden matrix, enabling payloads >700 mg per 2 mL syringe without enzymatic aids or major device redesigns. Yet it introduces a new particulate API modality, likely demanding more extensive particle characterization, leachables profiling, and particulate safety evaluation under regulatory scrutiny.
Risks, unknowns, and governance considerations
- In vivo performance: No published animal pharmacokinetics, efficacy, or toxicity data exist. Release kinetics, depot morphology, bioavailability, and immunogenicity profiles remain to be determined.
- Regulatory pathways: Particulate delivery of ASD API raises CMC questions: particle size distribution, residual pentanol levels, extractables/leachables, sterile processing, and endpoint release assays.
- Scale-up feasibility: While microfluidic units can be paralleled, industrial-scale throughput, inline cleaning, and process validation protocols must be established to satisfy GMP requirements.
- Clinical tolerability: Subcutaneous depots of 100 µm particles could elicit local reactogenicity—pain, induration, or tissue remodeling—requiring targeted preclinical and early-phase human studies.
- Long-term stability: Four-month refrigerated stability is a baseline; commercial shelf-life expectations (12–24 months) under stress and varied temperature excursions demand further investigation.
Implications for stakeholders and next-phase questions
For formulation scientists and translational teams, the emergence of a particulate ASD platform implies near-term needs to characterize dissolution profiles, membrane permeation, and protein integrity post-resuspension. Key questions include whether the ASD matrix alters antibody structure or binding function and how tunable the release kinetics are across different mAb constructs.
Regulatory affairs and CMC groups will likely need to define characterization strategies for residual solvent quantification, alginate cross-link density, particle uniformity, sterility assurance levels, and in-process controls for continuous microfluidic reactors. Open inquiries cover acceptable analytical methods and comparability frameworks versus existing liquid formulations.
Clinical development teams face decisions on designing animal PK/Tox studies that capture local depot effects and systemic exposure. Questions center on dosing schedules for sustained release versus bolus delivery, potential immunogenicity from alginate or ASD components, and strategies for monitoring local tolerability.
Manufacturing and process-engineering functions should evaluate microfluidic module throughput projections, scale-out versus scale-up trade-offs, cleaning-in-place (CIP) validations, and integration with upstream antibody production and downstream filling lines. Feasibility studies will inform capital investment decisions and potential partnerships with microfluidics specialists.
Business development and licensing teams may interpret this advance as an opportunity to secure rights for at-home subcutaneous formulations of existing high-dose oncology or rare-disease antibodies, contingent on positive preclinical proof-of-concept and clarified CMC pathways. Strategic positioning will depend on timing of animal data release and initial GMP model demonstrations.
Conclusion – a pivotal advance amid persistent unknowns
The MIT microfluidic dehydration method removes a central manufacturing hurdle—centrifugation—enabling concentrated, syringeable antibody formulations at ~360 mg/mL with favorable syringeability and short-term stability. This innovation sharpens the engineering case for high-dose subcutaneous therapy but pivots the spotlight onto in vivo validation, regulatory acceptance of particulate ASDs, and robust scale-up strategies. As the field moves toward infusion-free biologics, these open questions will define the pace and breadth of clinical and commercial adoption.