Transient Hepatocyte-Driven Thymic Signals Expand Aged Mice’s Naive T-Cell Pools, yet Human Translation Encounters Safety and Durability Gaps
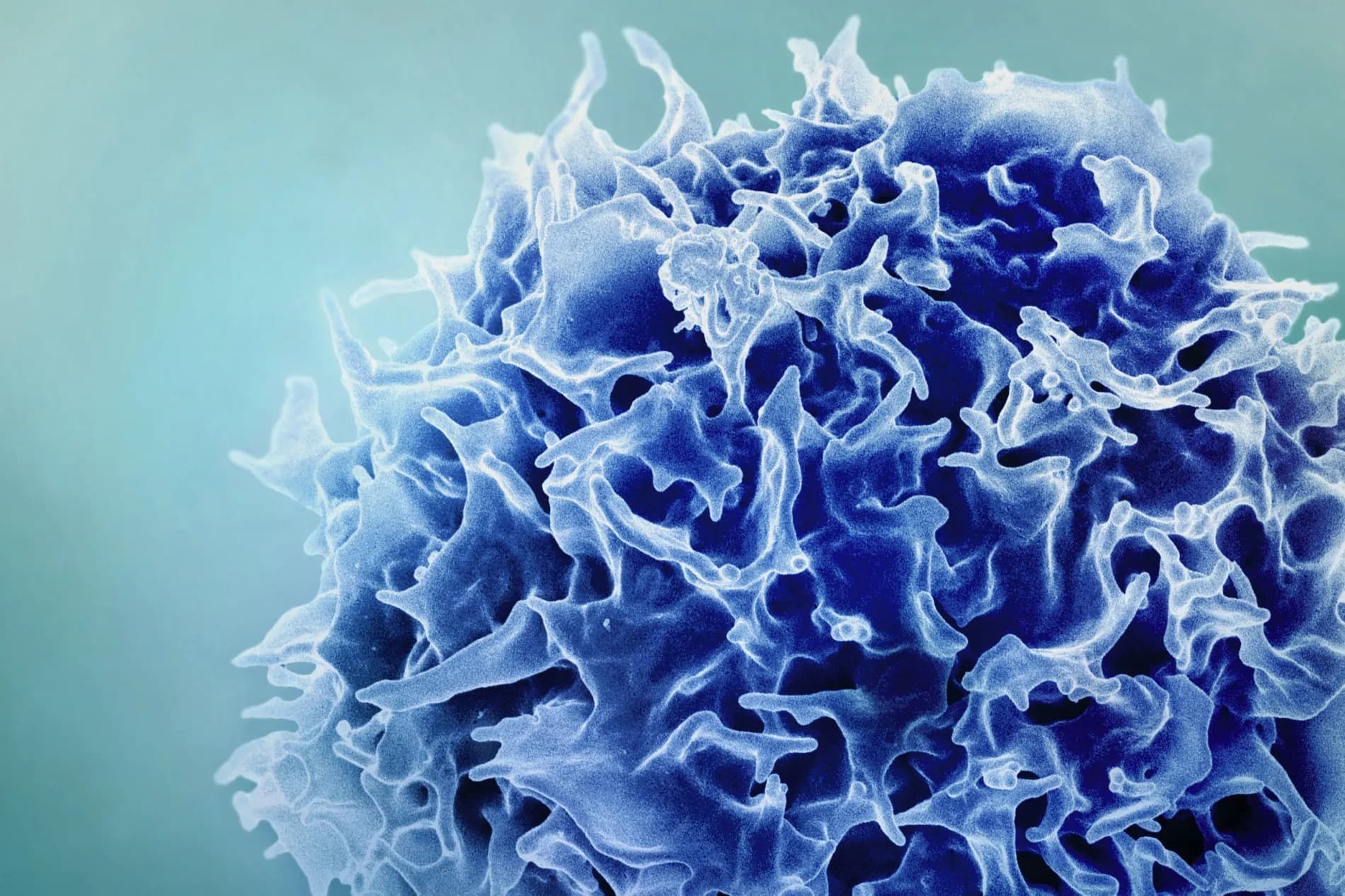
This study from MIT and the Broad Institute demonstrates that lipid-nanoparticle (LNP) mRNA encoding thymopoietic factors—DLL1, FLT3L and IL-7—can convert mouse hepatocytes into a temporary source of thymic signals, achieving a two- to threefold increase in splenic naive CD4+ and CD8+ T-cell numbers, enhanced T-cell receptor diversity and modestly improved vaccine and checkpoint-inhibitor responses in 18-month-old mice. While these findings delineate a novel organ-targeted route for immune rejuvenation in murine models, translation to humans remains speculative without non-human primate (NHP) toxicology, extended durability data beyond weeks and comprehensive safety profiling against off-target expression, hepatotoxicity and autoimmunity.
Mechanism and Key Preclinical Findings
The research encodes three thymic support proteins—Notch ligand DLL1, dendritic-cell growth factor FLT3L and the T-cell survival cytokine IL-7—into mRNA formulated in LNPs. Intravenous administration directs LNPs primarily to the liver, where hepatocytes transiently translate and secrete these factors. Over four weeks of dosing in aged mice, investigators observed:
- 2–3× expansion of splenic naive CD4+ and CD8+ T-cell populations measured by flow cytometry;
- Broadened T-cell receptor (TCR) repertoire diversity quantified via high-throughput sequencing;
- Enhanced antibody titers and recall responses in model vaccine challenges;
- Improved tumor control in a melanoma model treated concomitantly with anti-PD-1 therapy.
All effects were documented within murine timelines: naive T-cell counts returned toward baseline several weeks after dosing ceased, and functional gains in vaccine and tumor models were similarly transient.
Immunosenescence and the Thymic Involution Challenge
Thymic involution—the gradual shrinkage and functional decline of the thymus—drives reduced naive T-cell output, constricts TCR diversity and impairs adaptive immunity in aged organisms. In humans, diminished naive T-cell pools correlate with increased susceptibility to novel pathogens, reduced vaccine efficacy and poorer cancer immunotherapy outcomes. Conventional strategies include direct thymic regeneration via growth-hormone cocktails or cellular therapies targeting thymic epithelial cells. The LNP-mRNA liver proxy sidesteps direct thymic manipulation by offering a transient, transplant-free source of thymopoietic cues, reframing the organ-specific delivery paradigm.

Comparative Landscape of Immune-Rejuvenation Approaches
Two main categories have dominated preclinical and early clinical efforts:
- Systemic hormone or growth-factor regimens: Intervene Immune’s combination of growth hormone, DHEA and metformin has advanced into small human pilot studies aiming for epigenetic reversal and thymic tissue regrowth.
- Direct thymic modulators: Agents such as RANKL protein injections and TECregen’s ligand-engineered growth factors focus on rebuilding the thymic epithelial niche through repeated dosing or cell-engineering methods.
By contrast, the LNP-mRNA liver approach achieves systemic facilitation of naive T-cell maturation without requiring cellular transplantation or chronic systemic cytokine infusions. Its transient nature and reliance on mRNA manufacturing may simplify production compared to cell-therapy platforms, but it remains untested outside rodent models.
Durability, Safety Uncertainties and Extrapolation Limits
The murine data underscore a proof-of-concept, yet several critical unknowns temper anticipation of human efficacy:
- Durability measured in weeks: The restored naive T-cell pools began contracting within weeks post-treatment, raising questions about dosing frequency and long-term maintenance.
- Off-target expression risks: LNP biodistribution studies remain limited; unintended cytokine production in non-hepatic tissues could provoke systemic immune stimulation or autoimmunity.
- Hepatotoxicity concerns: Repeated LNP dosing has not been assessed in aged non-human primates, where liver physiology diverges from mice and may reveal tolerance limits or inflammatory responses.
- Autoimmunity potential: Rapid expansion of naive T cells without full peripheral tolerance checks could unmask autoreactive clones in an aged immune system.
Claims of human equivalence—such as equating 18-month-old mice to middle-aged adults—should be viewed as approximate, reflecting some parallels in immunosenescence models but not predictive of clinical outcomes. All functional readouts (vaccine seroconversion, tumor regression) derive from mouse frameworks; human immune complexity and longevity effects remain uncharted.
Regulatory and Translational Implications
The trajectory from rodent proof-of-concept to human therapy traditionally traverses NHP safety, biodistribution and toxicology studies, followed by phase 1 trials focused on tolerability and biomarker endpoints. In this context:
- Non-human primate toxicology will be essential to map LNP biodistribution beyond the liver and to monitor potential inflammatory or fibrotic liver changes with repeated dosing.
- Extended durability studies in larger animals could define minimal effective dosing intervals and inform manufacturing scale and cold-chain logistics.
- Early human trials would likely prioritize older adults with diminished vaccine responses, using naive T-cell expansion and TCR diversity as mechanistic biomarkers rather than direct clinical efficacy readouts.
- Safety monitoring must incorporate autoimmunity panels, liver-enzyme surveillance and independent safety oversight to guard against unforeseen off-target effects.
These translational steps remain speculative until peer-reviewed publication and replication of murine results. The absence of NHP or human data underscores that clinical readiness is currently aspirational rather than imminent.
Implications for Stakeholders
- For immunology researchers: The findings highlight a modular platform for organ-targeted cytokine delivery via mRNA, warranting exploration of alternative tissue proxies and dosing regimens to optimize tolerance and durability.
- For biomanufacturing teams: Scale-up of LNP-mRNA production must reconcile potential high-frequency dosing with manufacturing throughput, quality control for multiple protein-encoding transcripts and cold-chain distribution demands.
- For preclinical safety experts: Prioritizing comprehensive biodistribution analytics and dose-escalation studies in aged NHPs could clarify the hepatic safety profile and autoimmunity thresholds.
- For translational investors: The study demonstrates a new use case for mRNA beyond vaccines, yet valuation should account for significant translational risk given the preclinical-only status and anticipated investment in large-animal and early-phase human studies.
- For regulators: The approach challenges conventional classification of mRNA therapeutics, blending cytokine-based immunomodulation with tissue-targeted gene delivery, and will demand tailored guidelines on vector biodistribution and immune-stimulation safety.
Conclusion
By converting the liver into a fleeting thymic surrogate, LNP-mRNA encoding DLL1, FLT3L and IL-7 achieves measurable immune rejuvenation in aged mice, marking a conceptual advance in organ-targeted cytokine delivery. However, the path to human application hinges on resolving durability limitations, demonstrating safety in non-human primates and establishing robust regulatory frameworks for repeated mRNA dosing. Until those translational milestones are met, the technology remains a promising rodent innovation rather than a clinical reality.